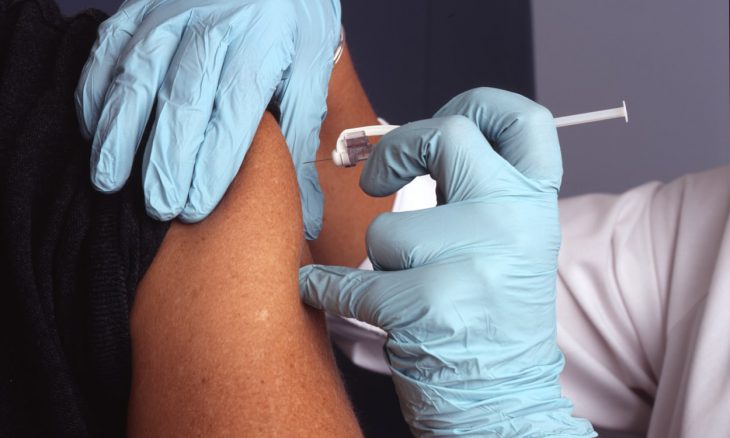
A serious adverse effect triggered pause in study.
Johnson & Johnson announced it was pausing shots in the late-stage coronavirus vaccine trial due to an adverse event, but said they will continue to enroll patients and stick to the current manufacturing timeline.
It is unclear if the participant was receiving a placebo or the vaccine, a point that will be determined by an independent industry group—the Data Safety Monitoring Board.
Another vaccine using similar technology from AstraZeneca remains on hold after a serious adverse effect from a trial participant triggered a halt in September. The trial has continued in the U.K. but remains on hold in the United States.
A statement from J&J said, “Adverse events—illnesses, accidents, etc.—even those that are serious, are an expected part of any clinical study, especially large studies.” The company promised a “careful review of all of the medical information before deciding whether to restart the study.”
Information on is kept confidential from the company until after the safety board reviews it, and it takes a few days at minimum for the right set of information to be gathered and evaluated.
Two other companies working on a vaccine, Moderna and Pfizer with BioNTech have so far not had any significant adverse events.
As the Lord Leads, Pray with Us…
- For the laboratories that are working hard on vaccines and therapies to address the COVID-19 virus.
- For protection for the people who have volunteered to be part of the trial groups to test the vaccines.
- That none of the “events” that have resulted in a pause in the study would be life-threatening to the participants.
Sources: Wall Street Journal, Washington Post